Principle of Secondary Battery(Lithium-ion battery)

Comparison of LFP and NCM Batteries
There are two main types of secondary batteries.
NCM batteries that are mainly made in Korea(Eco Pro, LG Energy, Samsung SDI)
LFP batteries made mainly in China(CATL, BYD)
NCM stands for Nickel, Cobalt, and Manganese, and it’s a type of lithium-ion battery that uses these materials. LFP batteries, on the other hand, use lithium iron phosphate.
However, the most significant difference between NCM and LFP lies in the energy density derived from the materials. Energy density refers to the amount of energy stored per unit weight, essentially the difference in the amount of electricity stored in the same 1kg battery.
NCM batteries have an energy density of around 305Wh/kg, whereas LFP batteries in China have an energy density of 165Wh/kg. This means that NCM batteries are approximately 85% more efficient and about 46% lighter than LFP batteries.
In summary, NCM batteries are high-performance batteries with a high energy density made by South Korean companies like SK Innovation, LG Energy Solution, and Samsung SDI.
When examining the operating principle of lithium-ion batteries made in South Korea, it can be summarized as follows

Discharge is not of great importance; the focus should be on charging. During charging, lithium ions move from the positive electrode to the negative electrode. The chemical process of lithium ions moving from the positive electrode to the negative electrode during charging is the force that charges the battery.
In this way, sending lithium ions from the positive electrode to the negative electrode generates electricity.
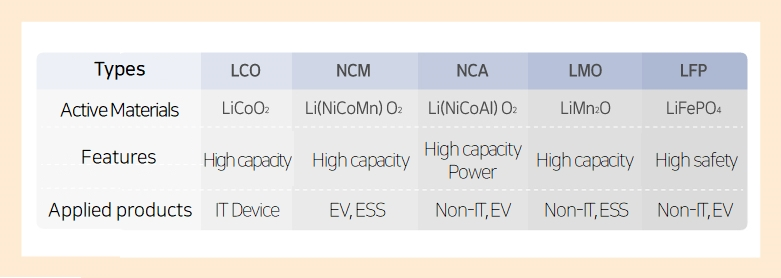
The definition and function of the positive electrode material
The correct term for “positive electrode material” is “Active material” in precise language. This Active material plays a role in providing lithium ions to the negative electrode during charging.
- Active material: An “Active material” or active material refers to a substance in the positive and negative electrodes of a battery that chemically reacts to generate electrical energy.
Materials such as lithium, nickel, cobalt, manganese, and others are used for this positive electrode material. Conceptually, it is a substance that facilitates the movement of ions between the positive and negative electrodes.
To summarize, a battery charges as lithium ions move from the positive electrode to the negative electrode.
This charging process determines the energy density of the battery.
Higher energy density allows for the production of lighter and higher-performance batteries, which significantly impacts battery performance.
The fundamental component responsible for this ion movement is the positive electrode material, which can be considered the heart of the battery.

3 thoughts on “Principles of secondary battery and anode materials”